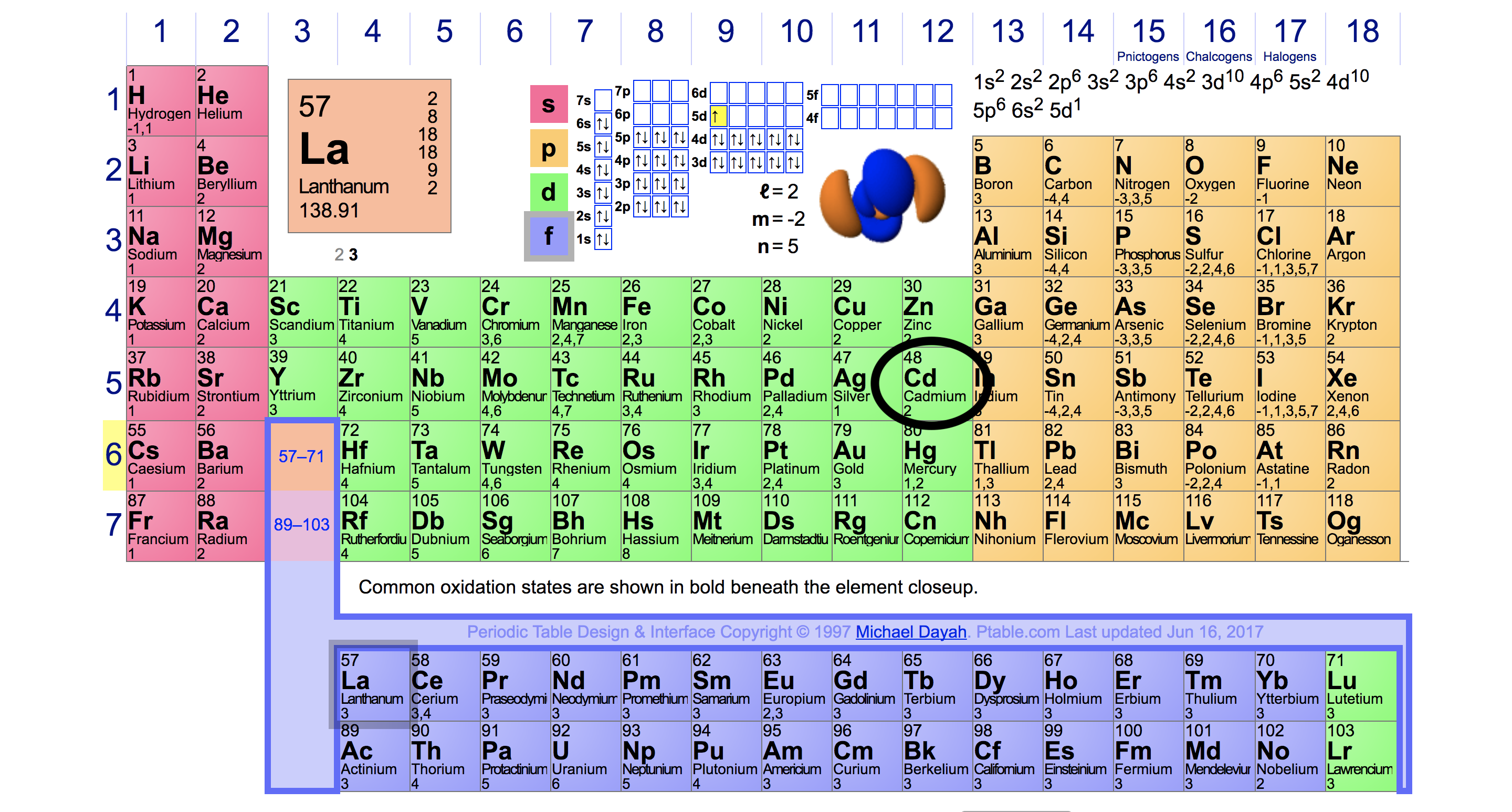
And in this third show we have our seven valence electrons, The remaining 10 electrons are in shells too, And one they total up to a total of electrons. And you can see here with this representation of the chlorine atom We have here are 3rd shell, so and equals three. So that would mean out of the 17 total electrons. Chlorine has an atomic number of 17 When it is neutral it has 17 protons. So you're gonna say total electrons Which is connected to your atomic number for neutral element equals your valence electrons plus your inner core electrons. We're going to say here that your total number of electrons. So these are the remaining electrons that are not valence electrons. Now, besides the valence electrons, all the remaining electrons are called your inner core electrons. Now these valence electrons are the outer shell electrons involved in forming chemical bonds. 9.9: Periodic Trends- Atomic Size, Ionization Energy, and Metallic Character Certain properties-notably atomic radius, ionization energy, electron affinity and metallic character-can be qualitatively understood by the positions of the elements on the periodic table.Now for main group elements were going to say that the number of valence electrons that they possess equals their group number.9.8: The Explanatory Power of the Quantum-Mechanical Model The chemical properties of elements is determined primarily by the number and distribution of valence electrons.

Electron configurations can be predicted by the position of an atom on the periodic table. 9.7: Electron Configurations and the Periodic Table The arrangement of electrons in atoms is responsible for the shape of the periodic table.Orbital filling diagrams are a way of indicating electron locations in orbitals. Hund's rule specifies the order of electron filling within a set of orbitals. The Aufbau principle gives the order of electron filling in an atom. Electron configuration notation simplifies the indication of where electrons are located in a specific atom. 9.6: Quantum-Mechanical Orbitals and Electron Configurations We look at the four quantum numbers for a given electron.Orbitals are mathematically derived regions of space with different probabilities of having an electron. This field deals with probabilities since we cannot definitely locate a particle. 9.5: The Quantum-Mechanical Model- Atoms with Orbitals Quantum mechanics involves the study of material at the atomic level.The state of electrons in atoms is described by four quantum numbers.